|
(by clicking the figure below or the link above). To further explore the organization of the Periodic Table and to become familiar with key patterns and trends, launch the TED-Ed clickable Periodic Table. Review the diagram below to become more familiar with how the periodic table is organized. Metals are found to the right of the staircase and non-metals are found to the left of the staircase metalloids are found to be touching the staircase and include. The Periodic Table is organized such that elements are arranged in groups that go vertically (up and down) and are numbered 1 - 18, while periods go horizontally (across) and are numbered 1 - 7. The Periodic Table of Elements is organized based on the physical properties (definition: a characteristic of a substance observed without creating a new substance can be qualitative or quantitative ) and chemical properties (definition: a description of what a substance does when it reacts to produce a new substance an ability or behaviour ) of the elements. Two non-adjacent elements in the same group (vertical column)įigure #3: A condensed periodic table showing the Bohr-Rutherford diagrams for the first 20 elements. Two non-adjacent elements in the same period (horizontal row) In your graphic organizer, record observations that compare and contrast (look for similarities and differences) the following: Let's consider what patterns and trends allow the periodic table to be organized the way it is: While reviewing the picture, complete the Exploration Framework to prompt your thinking and formulate inquiry-based questions. Review Figure #3 (below) that shows the Bohr-Rutherford diagrams for the first 20 elements of the periodic table.  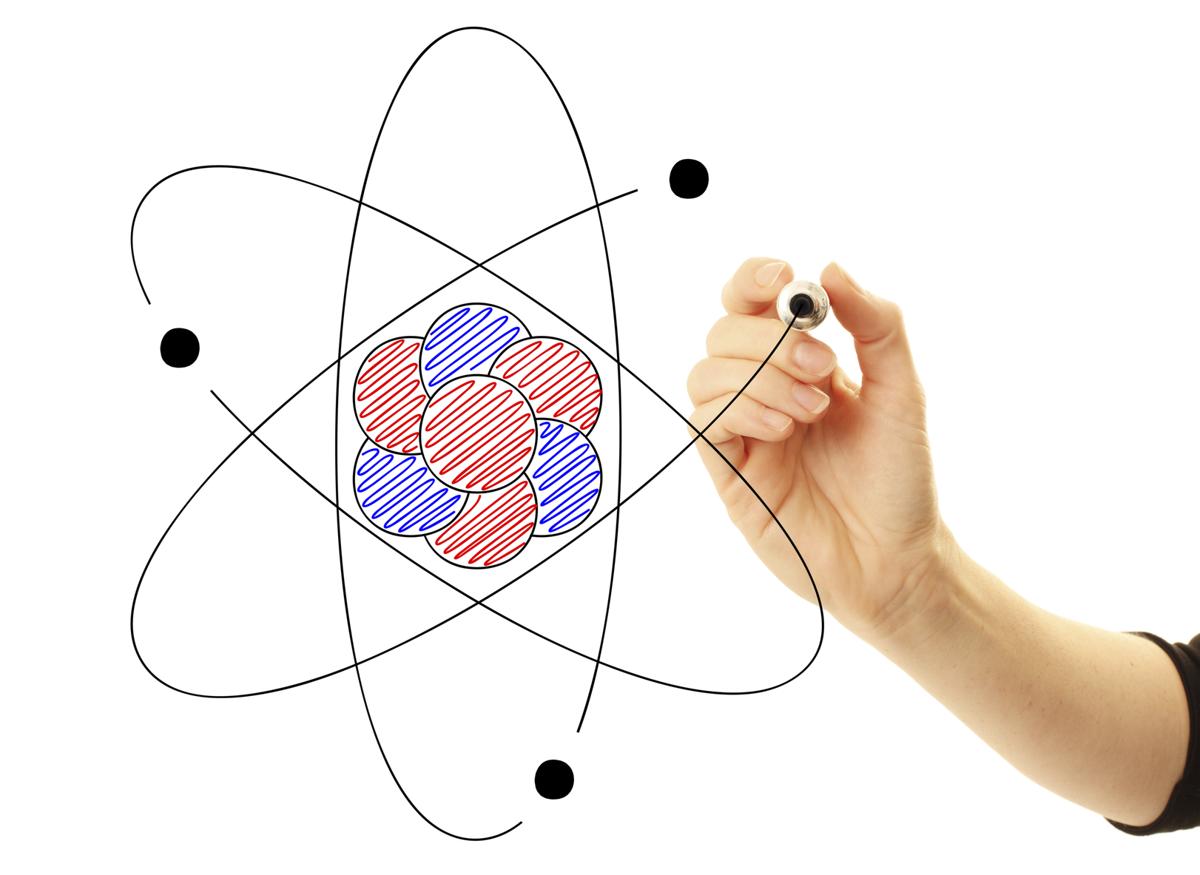
This movement results in a chemical change! (definition: A change in a substances that involves the formation of a new product that is different from the original consider the six clues to identify if a chemical change has occurred - energy produced, new colour, new smell, bubbles (gas), new solid forms, change is difficult to reverse. Valence electrons are those that are in the outermost energy shellĪ lot of emphases is placed on the outside electrons since these are the ones that move from one atom to another. Protons (p+) and neutrons (n0) are shown in the nucleus (centre)Įlectrons (red dots) are shown to orbit (move around) the nucleus in energy shells 
(definition: negatively charged subatomic particle found orbiting the nucleus )Īn example of a Bohr-Rutherford Diagram for oxygen is shown in Figure #2: While watching the video, use the Two Column Graphic Organizer to record your thoughts and ideas.īohr-Rutherford Diagrams & The Periodic Table Finding Patterns Figure #2: Bohr-Rutherford diagram for Oxygen.Ī Bohr-Rutherford diagram shows a visual representation of the atom (definition: smallest unit of that shows the chemical properties of an element made up of protons, neutrons, and electrons ) and accounts for the organization of its protons (definition: positively charged subatomic particle found in the nucleus ), neutrons (definition: neutral (no charge) subatomic particle found in the nucleus ), and electrons.Watch the video The Periodic Table Song from AsapSCIENCE: 
Scientists organize all elements on the periodic table. Remember that an individual atom is structured from three (3) subatomic particles called: (definition: smallest unit of that shows the chemical properties of an element ) In the previous activity you learned that the ‘stuff’ the universe is made of is called matter (definition: any material that has a mass and takes up space ) , which is made up of elements (definition: a substance that has specific chemical and physical properties and cannot be broken down to a smaller form through ordinary chemical reactions ). Of an atom is the sum of the protons and neutrons. Recall the Atomic Numberįor Helium is 2, giving the neutral atom 2 protonsĪnd 2 electrons. The Periodic Table Song! Figure #1: Model of a Helium atom Ĭenter (nucleus), while electrons (blue dots)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
